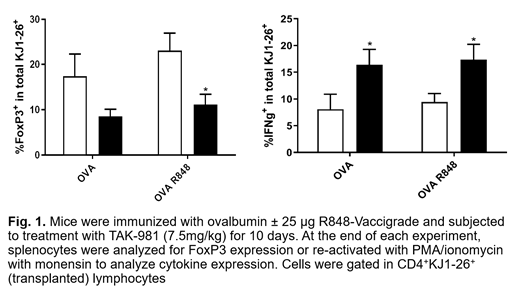Abstract
Introduction: CLL is characterized by deficient immunity which clinically manifests as increased predisposition towards malignancies and infectious complications. T-cells from patients with CLL exhibit a skewed repertoire with predominance of Tregs as well as impaired immune synapse formation and cytotoxic function. Small ubiquitin-like modifier (SUMO) family proteins regulate a variety of cellular processes, including nuclear trafficking, gene transcription and cell cycle progression, via post-translational modification of target proteins. Sumoylation regulates NFκB signaling, IFN response and NFAT activation, processes indispensable in immune cell activation. Despite this, the role of sumoylation in T cell biology in context of cancer is not known. TAK-981 is a small molecule inhibitor of the SUMO-activating enzyme (SAE) that forms a covalent adduct with an activated SUMO protein, thereby preventing its transfer to the SUMO-conjugating enzyme (Ubc9). Here, we investigated the immunomodulatory effects of TAK-981 in CLL.
Methods: T cells from patients with CLL were purified using Dynabeads. For polarization assays, FACS-sorted naïve CD4+ T cells were cultured for 7 days in control or differentiation media. For gene expression profiling (GEP; Clariom S), RNA was harvested after 3 and 24 hours of TCR engagement from FACS-sorted naïve CD4+ T cells. For in vivo immunization experiments, CD4+KJ1-26+ cells were inoculated IV into BALB/cJ mice. Mice received 100 µg IV ovalbumin ± R848 followed by TAK-981 7.5 mg/kg or vehicle control IV twice weekly for 10 days prior to spleen collection. Both recipient and transplanted splenocytes were analyzed. For analysis of tumor-infiltrating lymphocytes (TILs), BALB/c mice were injected with 1x10 6 A20 lymphoma cells and treated as above. TAK-981 was provided by Millennium Pharmaceuticals, Inc. (Cambridge, MA).
Results: T cells from patients with CLL demonstrated high baseline protein sumoylation that slightly increased following TCR engagement (αCD3/CD28). Treatment with TAK-981 significantly downregulated SUMO1 and SUMO2/3-modified protein levels yet did not disrupt early TCR signaling as evidenced by sustained ZAP70, p65/NFκB and NFAT activation detected by immunoblotting, immunocytochemistry and GEP. Treatment with TAK-981 resulted in dose-dependent upregulation of the early activation marker CD69 in CD4 + T cells following 72 and 96 hours of TCR stimulation vs. control. Meanwhile, expression of CD25, HLA-DR and CD40L was delayed in the presence of TAK-981. Interestingly, CD38, an IFN response target, was induced two-fold in TAK-981-treated cells after 24 hours and persisted at high levels at subsequent timepoints. T cell proliferation was reduced in the presence of high (1 μM) but not low/intermediate concentrations of TAK-981, accompanied by reduced S phase entry and decreased synthesis of IL-2. However, T cells did not undergo apoptosis under those conditions.
Targeting SAE in either control or Th1/Treg polarizing conditions facilitated an increase in IFNγ and loss of FoxP3 expression (accompanied by decreased IL-2/STAT5), suggesting a shift towards Th1 and away from Treg phenotype, respectively. GEP (Reactome, GSEA) confirmed a dramatically upregulated IFN response in TAK-981-treated CD4 + naïve T cells. Furthermore, targeting SAE enhanced degranulation (CD107a), IFNγ and perforin secretion in cytotoxic CD8+ T cells and potentiated T cell cytotoxicity in allogeneic assays with lymphoma cells (OCI-LY3, U2932) as targets.
Consistent with our in vitro data, OVA-stimulated transplanted transgenic KJ1-26+ splenocytes, as well as total CD4+ T cells from recipient mice treated with TAK-981 in vivo exhibited a significant reduction in expression of FoxP3 and an increased production of IFNγ (Figure 1). In the A20 syngeneic model, treatment with TAK-981 similarly downregulated FoxP3 expression in CD4+ TILs and induced IFNγ secretion in CD8+ TILs.
Conclusion. Using a combination of in vitro and in vivo experiments, we demonstrate that pharmacologic targeting of sumoylation with TAK-981 does not impair proximal TCR signaling in T cells obtained from patients with CLL, but leads to rebalancing toward healthy immune T cell subsets via induction of IFN response and downmodulation of Tregs. These data provide a strong rationale for continued investigation of TAK-981 in CLL and lymphoid malignancies.
Siddiqi: Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; BeiGene: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Pharmacyclics LLC, an AbbVie Company: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; TG Therapeutics: Research Funding; Kite Pharma: Membership on an entity's Board of Directors or advisory committees, Research Funding; Oncternal: Research Funding; Janssen: Speakers Bureau; AstraZeneca: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Celgene: Membership on an entity's Board of Directors or advisory committees; BMS: Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau. Danilov: SecuraBio: Research Funding; Bayer Oncology: Consultancy, Honoraria, Research Funding; Genentech: Consultancy, Honoraria, Research Funding; Takeda Oncology: Research Funding; TG Therapeutics: Consultancy, Research Funding; Rigel Pharm: Honoraria; Abbvie: Consultancy, Honoraria; Beigene: Consultancy, Honoraria; Pharmacyclics: Consultancy, Honoraria; Gilead Sciences: Research Funding; Bristol-Meyers-Squibb: Honoraria, Research Funding; Astra Zeneca: Consultancy, Honoraria, Research Funding.